Currently out of stock
Contact us >
Dycoxan is a white Oral Suspension for lambs and calves has been created to prevent scours caused by coccidiosis.
Channelle have created it using the same active ingredient as the popular Vecoxan brand.
In lambs Dycoxan is used for the prevention of coccidiosis caused by Eimeria crandallis and Eimeria ovinoidalis.
In calves Dycoxan is used for the prevention of coccidiosis caused by Eimeria bovis and Eimeria zuernii.
The easy to use drench comes in a bottle supplied with a spouted cap included.
Each ml contains: diclazuril 2.5mg, methyl parahydroxybenzoate (E218) 1.8mg, propyl parahydroxybenzoate (E216) 0.2mg.
What is coccidiosis?
Coccidiosis is a highly contagious disease that normally infects calves between one and six months of age but can also infect older cattle as well. Coccidiosis is caused by a single celled microscopic parasite that multiplies in the intestine of infected animals, the eggs are then passed through faeces onto the soil and grass where they are eaten by other animals which then become infected. Coccidiosisis still one of the most common and costly diseases a farmer can face on his farm.
What are the symptoms of Coccidiosis?
Common symptoms of Coccidiosis are diarrhoea, rough coat, loss of appetite, loss of weight and weakness in calves. In severe cases there will be blood and mucus present in the animals manure and if left untreated will lead to the animal's death.
Active Ingredients
diclazuril 2.5mg,
methyl parahydroxybenzoate (E218) 1.8mg,
propyl parahydroxybenzoate (E216) 0.2mg
Target Species
Lambs and Calves
Application Method
Oral SolutionDrench
Dycoxan is a preventative measure for use in herds and flocks that have a history of coccidosis. If there is no recent and confirmed history of clinical coccidiosis, the presence of coccidia in the flock or herd should be confirmed by faecal sampling prior to treatment
Withdrawal Period for Dycoxan
Dycoxan has a zero day withdrawal for both lambs and Calves
Dosage of Dycoxan
1 mg diclazuril per kg body weight
(i.e 1 ml of the oral suspension per 2.5 kg body weight),
in a single oral administration for both lambs and calves.
Read the package leaflet before use.
Features of Dycoxan
This Product is only licensed for sale in the Republic of Ireland
Click here to Download Data Sheet
Health Products Regulatory Authority
Summary of Product Characteristics
1 NAME OF THE VETERINARY MEDICINAL PRODUCT
Dycoxan 2.5 mg/ml Oral Suspension for sheep and cattle
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
1 ml contains
Active substance:
Diclazuril 2.5 mg
Excipients:
Methyl Parahydroxybenzoate (E218) 1.8 mg
Propyl Parahydroxybenzoate 0.2 mg
For the full list of excipients, see section 6.1
3 PHARMACEUTICAL FORM
Oral suspension.
White to off-white suspension.
4 CLINICAL PARTICULARS
4.1 Target Species
Sheep (lambs), Cattle (calves)
4.2 Indications for use, specifying the target species
In lambs:
Prevention of clinical signs of coccidiosis caused by Eimeria crandallis and Eimeria ovinoidalis sensitive to diclazuril. In calves:
Prevention of clinical signs of coccidiosis caused by Eimeria bovis and Eimeria zuernii sensitive to diclazuril.
4.3 Contraindications
Do not use in cases of hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings for each target species
Avoid under-dosing, which may be due to underestimation of body weight, misadministration of the product or lack of calibration of the dosing device (if any).
It is recommended to treat all lambs of the flock and all calves in a pen.
This will contribute to reduce the infection pressure and assure a better epidemiological control of the coccidiosis infection.
If there is no recent and confirmed history of clinical coccidiosis, the presence of coccidia in the flock or herd should be confirmed by faecal sampling prior to treatment.
In certain cases, only a transient reduction of oocyst shedding may be achieved.
Suspected clinical cases of resistance to anticoccidials should be further investigated and where evidence strongly suggest resistance to a particular antiprotozoal, an anticoccidial belonging to another pharmacological class and having a different mode of action should be used.
Frequent and repeated use of antiprotozoals may lead to the development of resistance in the target parasite.
4.5 Special precautions for use
Special precautions for use in animals
Lambs
On rare occasions, in highly susceptible lambs e.g. where they have been housed for long periods of time before being turned out onto heavily contaminated pasture, a severe scour has been seen shortly after dosing.
In such cases, fluid therapy is essential.
Calves
Clinical coccidiosis generally occurs late in the parasite’s life cycle after most of the damage to the calf’s intestine has already been done.
This severely damaged intestine can easily be infected by secondary bacteria and/or other agents.
In cases of acute clinical coccidiosis treated with the product, fluid therapy is essential.
Symptoms of clinical disease may remain obvious in some calves treated with the product, even though oocyst excretion is reduced to a very low level, and overall prevalence of diarrhoea is decreased.
The preferred timing of treatment is directed by the known epidemiology of Eimeria spp. and the presence of coccidia in the flock or herd should be confirmed by faecal sampling prior to treatment, if there is no recent and confirmed history of clinical coccidiosis.
Coccidiosis is an indicator of insufficient hygiene in the flock/pen.
It is recommended to improve hygiene and to treat all lambs in the flock and all calves in a pen.
Special precautions to be taken by the person administering the veterinary medicinal product to animals
Wash hands after use.
4.6 Adverse reactions (frequency and seriousness)
In very rare cases, adverse events involving gastrointestinal disorders (such as diarrhoea, with possible presence of blood), lethargy and/or neurological troubles (agitation, recumbency, paresis...) have been reported.
Some treated animals may show signs of clinical disease (diarrhoea) even though oocyst excretion is reduced to a very low level.
The frequency of adverse reactions is defined using the following convention:
- very common (more than 1 in 10 animals treated displaying adverse reaction(s))
- common (more than 1 but less than 10 animals in 100 animals treated)
- uncommon (more than 1 but less than 10 animals in 1,000 animals treated)
- rare (more than 1 but less than 10 animals in 10,000 animals treated)
- very rare (less than 1 animal in 10,000 animals treated, including isolated reports)
4.7 Use during pregnancy, lactation or lay
Not applicable.
4.8 Interaction with other medicinal products and other forms of interactions
None known.
4.9 Amounts to be administered and administration route
For oral use only.
To ensure the correct dosage, bodyweight should be determined as accurately as possible.
If animals are to be treated collectively rather than individually, they should be grouped according to their bodyweight and dosed accordingly, in order to avoid under- or overdosing.
1 mg diclazuril per kg bodyweight (equivalent to 1 ml of the oral suspension per 2.5 kg bodyweight) as a single oral administration.
Lambs: A single oral administration of 1 mg diclazuril per kg bodyweight or 1 ml the product oral suspension per 2.5 kg bodyweight at about 4-6 weeks of age at the time that coccidiosis can normally be expected on the farm.
Under conditions of high infection pressure, a second treatment may be indicated about 3 weeks after the first dosing.
Calves: A single administration of 1 mg diclazuril per kg bodyweight or 1 ml the product oral suspension per 2.5 kg bodyweight, administered as a single dose, 14 days after moving into a potentially high risk environment. To ensure the correct dosage, bodyweight should be determined as accurately as possible.
DOSAGE GUIDE:
Bodyweight (Lambs and Calves) Dose Volume 1 mg/kg
5.0 kg 2 ml
7.5 kg 3 ml
10.0 kg 4 ml
12.5 kg 5 ml
15.0 kg 6 ml
20.0 kg 8 ml
25.0 kg 10 ml
50.0 kg 20 ml
75.0 kg 30 ml
100.0 kg 40 ml
150.0 kg 60 ml
175.0 kg 70 ml
200.0 kg 80 ml
If animals are to be treated collectively rather than individually, they should be grouped according to their bodyweight and dosed accordingly, in order to avoid under- or overdosing.
If a satisfactory response is not observed, then further advice should be sought from your veterinary surgeon and the cause of the condition should be reviewed.
It is good practice to ensure the cleanliness of calf housing.
Method of administration
Shake well before use.
The product oral suspension should be administered with a drenching gun.
Appropriate drenching equipment should be used to allow accurate dosing.
This is particularly important when administering small volumes.
4.10 Overdose (symptoms, emergency procedures, antidotes), if necessary
Diclazuril oral suspension was given to lambs as a single dose up to 60 times the therapeutic dose.
No adverse clinical effects were reported.
No adverse effects were noted either at 5 times the therapeutic dose administered four consecutive times with a 7-day interval.
In calves,
the product was tolerated when administered up to five times the recommended dose rate.
4.11 Withdrawal period(s)
Meat and offal:
Sheep (lambs): zero days
Cattle (calves): zero days
5 PHARMACOLOGICAL or IMMUNOLOGICAL PROPERTIES
Pharmacotherapeutic group:
Antiprotozoals, triazine derivatives
ATC vet code:
QP51AJ03
5.1 Pharmacodynamic properties
Diclazuril is an anticoccidial of the benzene acetonitrile group and has an anticoccidial activity against Eimeria species. Depending on the coccidia species, diclazuril has a coccidiocidal effect on the asexual or sexual stages of the development cycle of the parasite.
Treatment with diclazuril causes interruption of the coccidial cycle and of excretion of oocysts for approximately 2 to 3 weeks after administration. This allows the lambs to bridge the period of decrease of maternal immunity (observed at approximately 4 weeks of age) and calves to reduce the infection pressure of their environment.
5.2 Pharmacokinetic particulars
The absorption of diclazuril when administered as an oral suspension to lambs and calves is poor.
In lambs, peak plasma concentrations are reached about 24 hours after dosing.
The absorption decreases with the age of the lambs.
The elimination half‑life is about 30 hours.
In calves, kinetic profiles have been studied after administration of a single dose of 5 mg diclazuril per kg body weight and after dosing for 3 consecutive days at respectively 1 mg, 3 mg and 5 mg diclazuril per kg body weight. Following the single dose of 5 mg peak plasma concentrations in the range of 21 to 75 ng/ml were reached after 8 to 24 hours.
Thereafter the concentrations decreased with a half‑life of 16 hours to concentrations below 10 ng/ml after 48 hours. Following the 3 consecutive daily doses of 1 mg diclazuril per kg body weight, mean peak plasma concentrations of 65.6 ng/ml were reached 10.5 hours after the last dose.
Thereafter the concentrations decreased with a half‑life of 22 hours.
The AUC 0‑96 h was 2127 h.ng/ml.
Comparison with the profiles obtained after the multiple doses indicated dose proportionality and linearity. The time to reach peak plasma concentrations and the subsequent depletion half‑life were independent of the dose.
In vitro studies in ovine and bovine hepatocytes indicated that the metabolic transformation of diclazuril is very limited, as was also observed for other species.
In vivo studies in a number of animal species have also demonstrated that diclazuril is not excreted and excreted virtually completely unchanged with the faeces.
5.3 Environmental properites
Diclazuril has been shown to be very persistent in soil.
6 PHARMACEUTICAL PARTICULARS
6.1 List of excipients
Methyl Parahydroxybenzoate (E218)
Propyl Parahydroxybenzoate
Microcrystalline Cellulose
Carmellose Sodium Polysorbate 20
Sodium Hydroxide
Purified water
6.2 Major incompatibilities
In the absence of compatibility studies, this veterinary medicinal product must not be mixed with other veterinary medicinal products.
6.3 Shelf-life
Shelf-life of the veterinary medicinal product as packaged for sale: 2 years.
Shelf-life after first opening the immediate packaging: 6 months
6.4 Special precautions for storage
This veterinary medicinal product does not require any special storage conditions.
6.5 Nature and composition of immediate packaging
200 ml, PET bottle with child resistant tamper evident HDPE cap with LDPE lining
1 litre, 2.5 litre and 5 litre high density polyethylene bottle with polypropylene tamper-evident cap with Alu seal
Each pack size will be marketed with one container in a carton.
Not all pack sizes may be marketed.
6.6 Special precautions for the disposal of unused veterinary medicinal products or waste materials derived from the use of such products
Any unused veterinary medicinal product or waste material derived from such a veterinary medicinal product should be disposed of in accordance with local requirements.
7 MARKETING AUTHORISATION HOLDER
Chanelle Pharmaceuticals Manufacturing Limited
Loughrea
Co. Galway
Ireland
8 MARKETING AUTHORISATION NUMBER(S)
VPA10987/122/001
9 DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 4th May 2018
10 DATE OF REVISION OF THE TEXT
September 2020
Drench correctly
Sheep Oral Drench
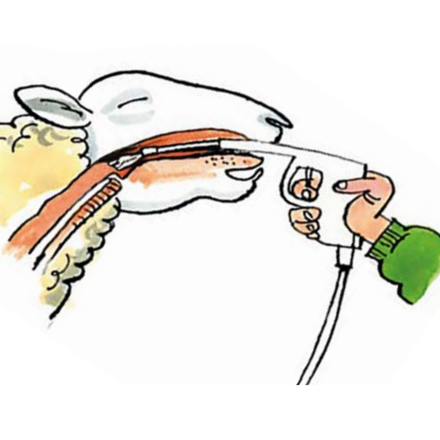
The drenching technique is a vital part of ensuring that the wormer does its job effectively.
Make sure the sheep are properly restrained and cannot leap around when they are being drenched so they swallow the whole amount.
Sheep can also suffer serious injury, or even death, if they are unrestrained and the gun penetrates the tissues at the back of the mouth.
Place a hand under the head and tilt slightly to the side.
Slot the nozzle in the gap between molar and incisor teeth and then over the back of the tongue.
If the wormer is just put into the mouth, it will bypass the rumen as it escapes down the oesophageal groove and will be less effective.
This is particularly important for white (BZ) drenches.
Dosing Weigh – do not guess Underestimating the weight of sheep is a common cause of underdosing. Select and weigh the biggest sheep in the group to determine the correct dose. If there is a wide range of weights, consider splitting the group, then weigh the heaviest in each section. Do not forget to check that the weigh crate is accurate before starting!
Calibrate and maintain the drench gun
Always check the gun is delivering the right amount before you drench. Remove the plunger from a 10 ml syringe, put a thumb over the end and squirt the dose into it, making sure there are no air bubbles left. Adjust the gun until the dose delivered is correct. Drenching guns should also be well maintained and replaced regularly. Clean with warm soapy water after use and check springs and tubes to make sure there are no kinks that will form air bubbles.
Withholding food
Research has shown that the efficacy of the white (BZ) and clear (AV) drenches can be improved by withholding food for 12–24 hours before treatment. It is not advised to deprive heavily pregnant ewes of food, so if you treat this class of stock with anthelmintics, you may wish to use yellow drenches (LV) because their efficacy is less dependent on rumen fill.
Storage
Wormers should be stored securely, away from direct sunlight at 4–25°C. Check the use-by date and, once open, use within the time shown on the packaging. Shake white (BZ) products well before use.
Cattle Oral drenches
Oral drenching guns are designed to deliver the treatment towards the back of the mouth over the tongue, so the entire dose is swallowed at once to optimise efficacy.
• Make sure animals are properly restrained, with their head held up
• Slide the nozzle of the dosing gun in the side of the mouth and over the tongue so that the entire dose is swallowed immediately
• Drenching equipment must be correctly calibrated and in good working order
• Calibrate the gun using the product just before treatment starts by delivering two or more doses into a graduated measuring cylinder.
Faulty equipment, or attempting to dose too quickly, may mean that the barrel of the gun does not fill properly or that the liquid is full of bubbles.

Here at Agridirect we have joined forces with DPD to ensure all packages are delivered promptly and safely to you. We ship to all mainland countries within the EU. Deliveries take place Monday to Friday excluding bank holidays. Once your order has been dispatched from our warehouse you will be notified by email. If there is a delay with your order for any reason you will be contacted immediately.
Due to Brexit we are temporarily unable to ship to the UK. Shipping to Northern Ireland will remain in place.
| Ireland (ROI & NI) | EU (Mainland Only) |
| 2-4 Working Days | 4-6 Working Days |
Some products have an extended delivery time, this is noted on the products.
| Country | Orders Under €129 | Orders Over €129 |
| Ireland (ROI & NI) | €7.99 | €0.00 |
| Austria | €24.99 | €17.00 |
| Belgium | €21.99 | €14.00 |
| Czech Republic | €24.99 | €17.00 |
| Denmark | €24.99 | €17.00 |
| Finland | €36.99 | €29.00 |
| France | €21.99 | €14.00 |
| Germany | €21.99 | €14.00 |
| Hungary | €28.99 | €21.00 |
| Italy | €34.99 | €23.00 |
| Luxenburg | €21.99 | €14.00 |
| Netherlands | €21.99 | €14.00 |
| Poland | €21.99 | €17.00 |
| Portugal | €36.99 | €29.00 |
| Slovakia | €28.99 | €21.00 |
| Slovenia | €28.99 | €21.00 |
| Spain | €34.99 | €27.00 |
| Sweden | €34.99 | €27.00 |
A selection of the products we sell are only licensed for sale within the Republic Of Ireland and can not be shipped outside of the country. These products are noted as only being available within the Republic of Ireland on the individual product pages.
There may be an addition charge on certain bulky items. This charge will be clearly marked on an applicable products and will be explained on the checkout page before payment has been made.
We’re sorry your purchase didn’t work out. But don’t worry; we have a great returns policy to help you out.
All purchases can be returned to us within 14 days of delivery and returned goods must be received within 14 days from the date you informed us of the return.
Purchases may be opened for inspection but must not be used and must be repackaged securely in the original packaging if you wish to return it.
If we discover goods have been used or there has been a loss in value of the goods due to damage to the goods, while in your care or whilst being returned to us, we will reduce the amount refunded, which may amount to the full cost of the product, to cover loss of value of goods.
All returns should be complete which includes boxes, manuals and accessories that may have been included with the order.
All returns must be packaged appropriately for shipping, we will not accept responsibility for damages or loss which occur during shipping of a return product.
We accept no responsibility for goods damaged or lost while in transit to us.
We have partnered with DPD to make your returns process easy and secure. simply follow the steps below and bring your package to an official DPD pickup point.
1) All returns must be accompanied with a fully filled out returns form which can be downloaded here.
2) To print off your return label click here or visit www.dpd.ie/returns and follow the on-screen instructions. Make sure and use your order number as your reference.
3) Bring your package to a DPD pickup point. To find your nearest drop off point here.
Once the returned product has been received into our warehouse and been fully inspected a refund will be issued.
If you choose not to use the DPD returns service we recommend that you use a method that can be tracked.
For the return of bulk products please contact us at sales@agridirect.ie
First off, if you have received a damaged electrical product from us, do not plug it in. Any electrical products that are plugged in are deemed ‘as used and accepted’ and are not accepted as returns. All damages must be reported to us via phone or email within 24hours of receipt of goods. Please ensure you check your items upon delivery.
How do I begin the returns process?
If you wish to begin the return process, please email us at sales@agridirect.ie and ensure the following information is included in your email. Your name, phone number, Order id, the item you wish to return, reason for return and if the product is damaged we require photos of the product.
Once you have sent us all required information a member of our team will assess your claim and will contact you as soon as possible. Please hold off on returning products until a member of our team has called you to confirm.
Once the returned product has been returned to us and fully inspected a refund will be issued.
Please Note: A typical timeline for a refund to show in your account is up to 10 working days from the date processed, depending on your bank.
Would you like to send this voucher to the recipient via email?
Yes No