This is a prescription-only medicine (POM).
A valid prescription is required to purchase this product.
Don’t have a prescription? Our team of vets is here to help.
Add to cart as normal and follow the steps. Available in Ireland only.
Volume:
Endofluke 100 mg/ml Oral Suspension is an orally administered drench used for the treatment of liver fluke in cattle and sheep. Endofluke 100 mg/ml fluke drench for cattle and sheep will treat mature, immature and early immature liver fluke (fasciola hepatica). Endofluke's active ingredient is triclabendazole which is the only active ingredient licensed in Ireland that has been proven to kill all three stages of liver fluke in sheep and cattle. Endofluke 100 mg/ml Oral Suspension has been approved for use in dairy cattle by the Irish Medicines Board (IMB).
Active Ingredient: Triclabendazole
Target Species: Cattle, sheep
Administration Method: Oral drench
Treats and Controls: Mature, immature and early immature liver fluke
Withdrawal Time: Animals should not be treated within 56 days of slaughter for human consumption. Cattle producing milk for human consumption need a withdrawl period of 48 hours after calving. Not intended for use within 45 days of calving. Should a cow calf earlier than 45 days after the last treatment, milk for human consumption may only be taken from 45 days plus 48 hours (47 days) after the last treatment. Not permitted for use in sheep producing milk for human consumption.
Dosage for cattle: 6 ml per 50 kg of bodyweight
| Body Weight | Dose Volume | Number of full doses per pack: | |
| 2.5 Litre | 5 Litre | ||
| 50kg | 6 ml | 416 | 833 |
| 100kg | 12 ml | 250 | 416 |
| 150kg | 18 ml | 138 | 277 |
| 200kg | 24 ml | 104 | 208 |
| 250kg | 30 ml | 83 | 166 |
| 300kg | 36 ml | 69 | 138 |
| 350kg | 42 ml | 59 | 119 |
| 400kg | 48 ml | 52 | 104 |
| 450kg | 54 ml | 46 | 92 |
| 500kg | 60 ml | 41 | 83 |
| 550kg | 66 ml | 37 | 75 |
| 600kg | 72 ml | 34 | 69 |
| Animals over 600 kg give an aditional 6 ml per 50 kg | |||
Dosage for sheep: 1ml per 10kg Bodyweight
| Dosage Rates For Sheep | |||
| Body Weight | Dose Volume | Number of full doses per pack: | |
| 2.5 Litre | 5 Litre | ||
| 10kg | 1ml | 2500 | 5000 |
| 20kg | 2 ml | 1250 | 2500 |
| 30kg | 3 ml | 833 | 1666 |
| 40kg | 4 ml | 625 | 1250 |
| 50kg | 5 ml | 500 | 1000 |
| 60kg | 6 ml | 416 | 833 |
| 70kg | 7 ml | 357 | 714 |
| Animals over 70 kg give an additional 1 ml per 10 kg | |||
Always read the label and all enclosed information for Endofluke Oral Suspension before administering to animals!
What are the effects of liver fluke infection?
This product is only licensed for sale within the Republic of Ireland
Click here to Download Data Sheet
Summary of Product Characteristics
1 NAME OF THE VETERINARY MEDICINAL PRODUCT
Endofluke 100 mg/ml Oral Suspension
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
Active Substance per ml
Triclabendazole 100mg
Excipients
Methyl Parahydroxybenzoate(E218) 2mg (preservative)
Propyl Parahydroxybenzoate (E216) 0.2mg (preservative)
For a full list of excipients, see section 6.1.
3 PHARMACEUTICAL FORM
Oral Suspension.
A white to off-white suspension.
4 CLINICAL PARTICULARS
4.1 Target Species
Cattle & Sheep
4.2 Indications for use, specifying the target species
For the treatment of adult, immature and early immature stages of liver fluke (Fasciola hepatica) susceptible to
triclabendazole.
4.3 Contraindications
Do not use in animals known to be hypersensitive to the active substance.
4.4 Special warnings for each target species
Care should be taken to avoid the following practices, because they increase the risk of development of resistance and could ultimately result in ineffective therapy:
- Too frequent and repeated use of anthelmintics from the same class, over an extended period of time.
- Under dosing, which may be due to under estimation of bodyweight, misadministration of the product or lack of
calibration of the dosing device (if any).
Suspected clinical cases of resistance to anthelmintics should be further investigated using appropriate tests (e.g. Faecal Egg Count Reduction Test). Where the results of the test(s) strongly suggest resistance to a particular anthelmintic, an anthelmintic belonging to another pharmacological class and having a different mode of action should be used.
Resistance to triclabendazole has been reported in Fasciola hepatica in cattle and sheep.
Therefore, the use of this product should be based on local (regional/farm) epidemiological information about susceptibility of the Fasciola hepatica and recommendations on how to limit further selection for resistance to anthelmintics.
4.5 Special precautions for use
i) Special precautions for use in animals
Care should be taken when dosing animals to avoid causing injury to the mouth and pharynx.
ii) Special Precautions to be taken by the person administering the product to animals
When using, do not eat, drink or smoke.
Wash splashes from eyes and skin immediately.
Take off immediately any contaminated clothing.
Wash hands and exposed skin before meals and after use.
iii) Other precautions
The use of this product may have harmful effects on fish and aquatic invertebrates. Cattle and
sheep must not have any access to the surface water such as streams, ponds or ditches within 7
days after treatment. When spreading manure from treated animals on arable lands, a safety distance of 10m to adjacent surface waters must be kept.
4.6 Adverse reactions (frequency and seriousness)
Occasionally, inflammation of the unpigmented skin, including the udder and the teats may occur after treatment in
cattle exposed to intense sunshine.
4.7 Use during pregnancy, lactation or lay
The product is safe for use during pregnancy and lactation.
However, the product is not permitted for use during lactation in animals producing milk for human consumption.
4.8 Interaction with other medicinal products and other forms of interaction
None known
4.9 Amounts to be administered and administration route
For single oral administration only using properly calibrated dosing equipment.
The product is suitable for most types of automatic drenching guns.
Shake the container before use.
Use unaltered from original container.
Clean drenching equipment before and after use.
Dosage:
Endofluke 100 mg/ml is given as an oral drench and is suitable for most types of automatic drenching guns.
The recommended dose rate is 12 mg triclabendazole per kg bodyweight in cattle and
10 mg triclabendazole per kg bodyweight in sheep.
Practical Dosage Guide:
Cattle: 6 ml per 50 kg bodyweight
Sheep: 1 ml per 10kg bodyweight
To ensure administration of a correct dose, bodyweight should be determined as accurately as possible; accuracy of the dosing device should be checked.
If animals are to be treated collectively rather than individually, they should be grouped according to their bodyweight
and dosed accordingly, in order to avoid under- or overdosing.
The timing for re-treatment should be based on epidemiological risk patterns and should be customised for each
individual farm.
To avoid the potential for the accumulation of residues following repeat administration of the product; animals should
not be treated with a frequency of less than 10 weeks.
|
AnimalWeight Dose of product
50 kg 6 ml
100 kg 12 ml
150 kg 18 ml
200 kg 24 ml
250 kg 30 ml
300 kg 36 ml
350 kg 42 ml
400 kg 48 ml
For each additional 50 kg 6 ml
AnimalWeight Dose of product
10 kg 1 ml
20 kg 2 ml
30 kg 3 ml
40 kg 4 ml
50 kg 5 ml
60 kg 6 ml
For each additional 10 kg 1 ml
4.10 Overdose (symptoms, emergency procedures, antidotes), if necessary
A single oral dose of 150-200 mg triclabendazole/kg of live bodyweight may lead to side effects such as unsteady gait, dullness and reduced appetite. These side effects are slight and last 1 to 5 days.
An antidote is not known.
4.11 Withdrawal Period(s)
Cattle (meat and offal): 56 days
Milk:
Cattle (milk): Milk for human consumption may only be taken from 48 hours after calving.
Not intended for use within 45 days of calving.
Should a cow calve earlier than 45 days after the last treatment, milk for human consumption mayonly be taken from 45 days + 48 hours (47 days) after the last treatment.
Sheep (meat & offal): 56 days
Sheep (milk): Not authorised for use in ewes producing milk for human consumption including during the dry period.
Do not use within 1 year prior to the first lambing in ewes intended to produce milk for human consumption.
5 PHARMACOLOGICAL or IMMUNOLOGICAL PROPERTIES
ATCvet code:
QP52AC01
Pharmacotherapeutic group:
anthelmintics; benzimidazoles and related substances.
5.1 Pharmacodynamic properties
Triclabendazole differs from other benzimidazoles in that it is a narrow spectrum anthelmintic. The drug accumulates
significantly in both immature and adult stages of Fasciola hepatica and stimulates the major routes of the parasite’s
energy generating system, as demonstrated by glucose derived acetate and propionate formation.
However, under these conditions the parasite’s motility decreased, indicating that the drug is not associated with inhibition of the energy generating pathways.
Triclabendazole inhibits colchicine binding to microtubular proteins suggesting interference of the drug with microtubular structure and function.
The drug strongly inhibits the release of proteolytic enzymes in immature and adult parasites, a process dependant on microtubular functions.
The precise molecular mode of action of this fasciolicidal drug remains to be elucidated.
5.2 Pharmacokinetic properties
50-75% of the orally administered dose of triclabendazole is absorbed from the gastrointestinal tract.
Very rapidly, absorbed triclabendazole is almost completely oxidised to its sulfoxide and sulfone.
In cattle triclabendazole sulfoxide reaches peak concentrations approximately 27 hours after administration of the
product and the sulfone reaches peak concentrations 64 to 72 hours after administration.
In sheep triclabendazole sulfoxide reaches peak concentrations approximately 20 hours after administration of the
product and the sulfone reaches peak concentrations 30 to 32 hours after administration.
Both metabolites bind strongly to plasma proteins, particularly albumin.
Metabolites are excreted via the bile mainly as conjugates. More than 90%- 95% of the total dose of triclabendazole is excreted in the faeces, about 2% in the urine and less than 1% in the milk.
The elimination is virtually complete by 10 days after administration.
6 PHARMACEUTICAL PARTICULARS
6.1 List of excipients
Xanthan Gum
Methyl Parahydroxybenzoate
Propyl Parahydroxybenzoate
Citric Acid Anhydrous
Sodium Citrate
Polysorbate 80
Silica Colloidal
Anhydrous Simethicone Emulsion
Water, purified
6.2 Incompatibilities
None Known.
6.3 Shelf-life
Shelf life of the veterinary medicinal product as packaged for sale: 2 years
6.4 Special precautions for storage
Protect from frost.
6.5 Nature and composition of immediate packaging
1 litre, 2.5 and 5 litre high-density polyethylene flat bottom backpack containers containing a white to off-white smooth
suspension.
The closures used for all pack sizes are 38 mm propylene standard caps with an induction heat seal liner.
A 38 mm polypropylene spouted cap is also provided with each pack size for dispensing purposes.
6.6 Special precautions for the disposal of unused veterinary medicinal products or waste materials
The product may have toxic effects on fish and aquatic invertebrates. Any unused product or waste material must not
enter surface water and should be disposed of in accordance with national requirements
7 MARKETING AUTHORISATION HOLDER
Cross Vetpharm Group Ltd.
Broomhill Road,
Tallaght,
Dublin 24.
8 MARKETING AUTHORISATION NUMBER(S)
VPA 10960/046/001
9 DATE OF THE FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 1st August 2002
Date of last renewal: 19th February 2008
10 DATE OF REVISION OF THE TEXT
November 2013
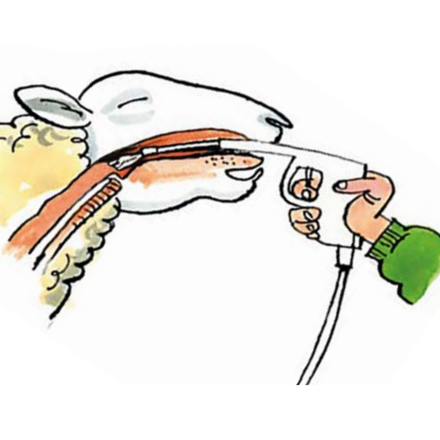
Drench correctly
Sheep Oral Drench
The drenching technique is a vital part of ensuring that the wormer does its job effectively.
Make sure the sheep are properly restrained and cannot leap around when they are being drenched so they swallow the whole amount.
Sheep can also suffer serious injury, or even death, if they are unrestrained and the gun penetrates the tissues at the back of the mouth.
Place a hand under the head and tilt slightly to the side.
Slot the nozzle in the gap between molar and incisor teeth and then over the back of the tongue.
If the wormer is just put into the mouth, it will bypass the rumen as it escapes down the oesophageal groove and will be less effective.
This is particularly important for white (BZ) drenches.
Dosing Weight – do not guess. Underestimating the weight of sheep is a common cause of underdosing. Select and weigh the biggest sheep in the group to determine the correct dose. If there is a wide range of weights, consider splitting the group, then weigh the heaviest in each section. Do not forget to check that the weigh crate is accurate before starting!
Calibrate and maintain the drench gun
Always check the gun is delivering the right amount before you drench. Remove the plunger from a 10 ml syringe, put a thumb over the end and squirt the dose into it, making sure there are no air bubbles left. Adjust the gun until the dose delivered is correct. Drenching guns should also be well maintained and replaced regularly. Clean with warm soapy water after use and check springs and tubes to make sure there are no kinks that will form air bubbles.
Withholding food
Research has shown that the efficacy of the white (BZ) and clear (AV) drenches can be improved by withholding food for 12–24 hours before treatment. It is not advised to deprive heavily pregnant ewes of food, so if you treat this class of stock with anthelmintics, you may wish to use yellow drenches (LV) because their efficacy is less dependent on rumen fill.
Storage
Wormers should be stored securely, away from direct sunlight at 4–25°C. Check the use-by date and, once open, use within the time shown on the packaging. Shake white (BZ) products well before use.
Cattle Oral drenches
Oral drenching guns are designed to deliver the treatment towards the back of the mouth over the tongue, so the entire dose is swallowed at once to optimise efficacy.
• Make sure animals are properly restrained, with their head held up
• Slide the nozzle of the dosing gun in the side of the mouth and over the tongue so that the entire dose is swallowed immediately
• Drenching equipment must be correctly calibrated and in good working order
• Calibrate the gun using the product just before treatment starts by delivering two or more doses into a graduated measuring cylinder.
Faulty equipment, or attempting to dose too quickly, may mean that the barrel of the gun does not fill properly or that the liquid is full of bubbles.

Pharmacodynamics
Triclabendazole and its metabolites are active against both the immature and mature worms of Fasciola hepatica and Fasciola gigantica helminths.
Mechanism of action
Triclabendazole is an anthelmintic agent against Fasciola species.
The mechanism of action against Fasciola species is not fully understood at this time.
In vitro studies and animal studies suggest that triclabendazole and its active metabolites (sulfoxide and sulfone) are absorbed by the outer body covering of the immature and mature worms, causing a reduction in the resting membrane potential, the inhibition of tubulin function as well as protein and enzyme synthesis necessary for survival. These metabolic disturbances lead to an inhibition of motility, disruption of the worm outer surface, in addition to the inhibition of spermatogenesis and egg/embryonic cells.
A note on resistance
In vitro studies, in vivo studies, as well as case reports suggest a possibility for the development of resistance to triclabendazole.
The mechanism of resistance may be multifactorial and include changes in drug uptake/efflux mechanisms, target molecules, and changes in drug metabolism.
Endofluke 100 mg/ml Oral Suspension is used for the treatment of liver fluke in cattle and sheep. Endofluke 100 mg/ml fluke drench for cattle and sheep will treat mature, immature and early immature liver fluke (fasciola hepatica).
Active Ingredient: Triclabendazole
Target Species: Cattle, sheep
Treats and Controls: Mature, immature and early immature liver fluke
Withdrawal Time: Animals should not be treated within 56 days of slaughter for human consumption. Cattle producing milk for human consumption need a withdrawl period of 48 hours after calving. Not intended for use within 45 days of calving. Should a cow calf earlier than 45 days after the last treatment, milk for human consumption may only be taken from 45 days plus 48 hours (47 days) after the last treatment. Not permitted for use in sheep producing milk for human consumption.
Legal Status: POM
Note: Some products may have an extra heavy/bulk delivery charge; this will be noted on the product page.
Here at Agridirect we have joined forces with An Post & DPD to ensure all packages are delivered promptly and safely to you. We ship to all mainland countries within the EU. Deliveries take place Monday to Friday, excluding bank holidays. Once your order has been dispatched from our warehouse, you will be notified by email. If there is a delay with your order for any reason, you will be contacted immediately.
| Ireland (ROI & NI) | EU (Mainland Only) |
| 2-4 Working Days | 4-6 Working Days |
Some products have an extended delivery time, this is noted on the products.
| Country | Orders Under €129 | Orders Over €129 |
| Ireland (ROI & NI) | €7.99 | €0.00 |
| Austria | €39.99 | €32.00 |
| Belgium | €36.99 | €29.00 |
| Czech Republic | €39.99 | €32.00 |
| Denmark | €39.99 | €32.00 |
| Finland | €51.99 | €45.00 |
| France | €36.99 | €29.00 |
| Germany | €36.99 | €29.00 |
| Hungary | €43.99 | €37.00 |
| Italy | €49.99 | €42.00 |
| Luxenburg | €36.99 | €29.00 |
| Netherlands | €36.99 | €29.00 |
| Poland | €36.99 | €29.00 |
| Portugal | €51.99 | €45.00 |
| Slovakia | €43.99 | €37.00 |
| Slovenia | €43.99 | €37.00 |
| Spain | €49.99 | €42.00 |
| Sweden | €49.99 | €42.00 |
A selection of the products we sell are only licensed for sale within the Republic Of Ireland and can not be shipped outside of the country. These products are noted as only being available within the Republic of Ireland on the individual product pages.
Note: There may be an addition charge on certain bulky items. This charge will be clearly marked on an applicable products and will be explained on the checkout page before payment has been made.
We’re sorry your purchase didn’t work out. But don’t worry; we have a great returns policy to help you out.
All purchases can be returned to us within 14 days of delivery, and returned goods must be received within 14 days from the date you informed us of the return.
Purchases may be opened for inspection but must not be used and must be repackaged securely in the original packaging if you wish to return it.
If we discover goods have been used or there has been a loss in value of the goods due to damage to the goods while in your care or whilst being returned to us, we will reduce the amount refunded, which may amount to the full cost of the product, to cover the loss of value of the goods.
All returns should be complete, which includes boxes, manuals, and accessories that may have been included with the order.
All returns must be packaged appropriately for shipping; we will not accept responsibility for damages or loss that occurs during shipping of a return product.
We accept no responsibility for goods damaged or lost while in transit to us.
We have partnered with DPD to make your returns process easy and secure. Simply follow the steps below and bring your package to an official DPD pickup point.
1) All returns must be accompanied with a fully filled out returns form which can be downloaded returns form PDF.
2) To print off your return label click DPD Returns page or visit www.dpd.ie/returns and follow the on-screen instructions. Make sure and use your order number as your reference.
3) Bring your package to a DPD pickup point. To find your nearest drop off point DPD pickup shops.
Once the returned product has been received into our warehouse and been fully inspected a refund will be issued.
If you choose not to use the DPD returns service we recommend that you use a method that can be tracked.
For the return of bulk products please contact us at sales@agridirect.ie
First off, if you have received a damaged electrical product from us, do not plug it in. Any electrical products that are plugged in are deemed ‘as used and accepted’ and are not accepted as returns. All damages must be reported to us via phone or email within 24hours of receipt of goods. Please ensure you check your items upon delivery.
How do I begin the returns process?
If you wish to begin the return process, please email us at sales@agridirect.ie and ensure the following information is included in your email. Your name, phone number, Order id, the item you wish to return, reason for return and if the product is damaged we require photos of the product.
Once you have sent us all required information a member of our team will assess your claim and will contact you as soon as possible. Please hold off on returning products until a member of our team has called you to confirm.
Once the returned product has been returned to us and fully inspected a refund will be issued.
Please Note: A typical timeline for a refund to show in your account is up to 10 working days from the date processed, depending on your bank.
Would you like to send this voucher to the recipient via email?
Yes No