The Most Common Equine Parasites And The Damage They Cause.
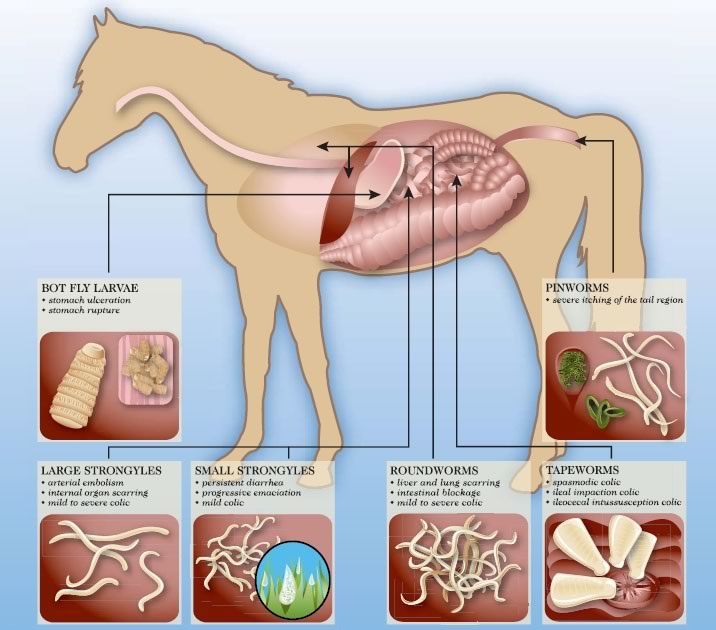
Use the diagram or the parasite menu to learn more about each parasite.
This is a prescription-only medicine (POM).
A valid prescription is required to purchase this product.
Don’t have a prescription? Our team of vets is here to help.
Add to cart as normal and follow the steps. Available in Ireland only.
Please Note: This product is for equine (horse) use only. It is not suitable for human consumption. Always follow veterinary advice.
Eqvalan Duo Horse Wormer (Ivermectin, Praziquantel) is an oral paste wormer for horses containing ivermectin and Praziquantel as it's active ingredients. Eqvalan Duo Horse Wormer treats a range of internal parasites, including small and large redworms, lungworms, pinworms and bots in horses.
Active Ingredient: Ivermectin, Praziquantel
Target Species: Horse
Treats and Controls: Large redworms, Small redworms, Pinworms, Lungworms, Ascarids, Stomach worms, Intestinal threadworms, Cutaneous worms, Bots
Administration Method: Oral paste
Withdrawal Time: Withdrawal period for meat and offal is 30 days. Do not use in mares producing milk for human consumption.
Dosage for Horses: Each syringe is sufficient to dose a horse up to 600kg.
Always read the label and all enclosed information for Eqvalan duo horse wormer before administering to animals!
Use the diagram or the parasite menu to learn more about each parasite.

Click here to Download Data Sheet
Health Products Regulatory Authority
Summary of Product Characteristics
1 NAME OF THE VETERINARY MEDICINAL PRODUCT
EQVALAN DUO,
oral paste
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
Each g contains:
Active substances:
Ivermectin 15.5 mg
Praziquantel 77.5 mg
Excipients:
Buthylhydroxyanisol (E320): 0.20 mg
Sunset Yellow (E110): 0.40 mg
Titanium dioxide (E171): 20mg
For the full list of excipients, see section 6.1.
3 PHARMACEUTICAL FORM
Oral paste.
Smooth, homogeneous orange paste.
4 CLINICAL PARTICULARS
4.1 Target Species
Horses.
4.2 Indications for use, specifying the target species
For the treatment of mixed cestode and nematode or arthropod infestations in horses.
The following parasites of horses are sensitive to the antiparasitic effects of EQVALAN DUO, oral paste:
Adult Tapeworms:
Anoplocephala perfoliata
Anoplocephala magna
Large strongyles:
Strongylus vulgaris (adults and arterial larval stages)
Strongylus edentatus (adults and tissue larval stages)
Strongylus equinus (adults)
Triodontophorus spp(adults)
Triodontophorus brevicauda
Triodontophorus serratus
Craterostomum acuticaudatum (adults)
Adult and immature (intraluminal fourth-stage larvae) of small strongyles or cyathostomes, including benzimidazole-resistant strains:
Coronocyclus spp
Coronocyclus coronatus
Coronocyclus labiatus
Coronocyclus labratus
Cyathostomum spp
Cyathostomum catinatum
Cyathostomum pateratum
Cylicocyclus spp
Cylicocyclus ashworthi
Cylicocyclus elongatus
Cylicocyclus insigne
Cylicocyclus leptostomum
Cylicocyclus nassatus
Cylicodontophorus spp
Cylicodontophorus bicornatus
Cylicostephanus spp
Cylicostephanus calicatus
Cylicostephanus goldi
Cylicostephanus longibursatus
Cylicostephanus minutus
Parapoteriostomum spp
Parapoteriostomum mettami
Petrovinema spp
Petrovinema poculatum
Poteriostomum spp
Adult hairworms: Trichostrongylus axei
Adult and immature (fourth stage Larvae) pinworms: Oxyuris equi
Adult, third- and fourth-stage larvae of roundworms (ascarids): Parascaris equorum
Microfilariae of neck threadworms: Onchocerca spp
Adult intestinal threadworms: Strongyloides westeri
Adult large-mouth stomach worms: Habronema muscae
Oral and, gastric stages of bots: Gasterophilus spp
Adult and immature (inhibited fourth stage larvae) lungworms: Dictyocaulus arnfieldi
4.3 Contraindications
Do not use in mares producing milk for human consumption.
The product has been formulated for use in horses only.
Cats, Dogs, especially Collies, Old English Sheepdogs and related breeds or crosses, and also turtles and tortoises may be adversely affected by the concentration of ivermectin in this product if they are allowed to ingest spilled paste or have access to used syringes
4.4 Special warnings for each target species
Care should be taken to avoid the following practices because they increase the risk of development of resistance and could ultimately result in ineffective therapy:
-Too frequent and repeated use of anthelmintics from the same class, over an extended period of time.
-Underdosing, which may be due to underestimation of body weight, misadministration of the product, or lack of calibration of the dosing device (if any).
Suspected clinical cases of resistance to anthelmintics should be further investigated using appropriate tests (e.g. Faecal Egg Count Reduction Test). Where the results of the test(s) strongly suggest resistance to a particular anthelmintic, an anthelmintic belonging to another pharmacological class and having a different mode of action should be used.
Resistance to macrocyclic lactones (which includes ivermectin) has been reported in Parascaris equorum in horses in the EU. Therefore the use of this product should be based on local (regional, farm) epidemiological information about susceptibility of gastro-intestinal nematodes and recommendations on how to limit further selection for resistance to anthelmintics
4.5 Special precautions for use
Special precautions for use in animals
Safety studies were not conducted in foals younger than 2 months of age, or in stallions, the use of Eqvalan Duo, oral paste is not recommended in these categories of animals.
Special precautions to be taken by the person administering the veterinary medicinal product to animals Wash hands after use.
Do not smoke, drink or eat while handling the product.
This product may cause skin and eye irritation.
Therefore, the user should avoid contact of the product with the skin and the eyes. In case of contact, rinse immediately with plenty of water.
In the case of accidental ingestion or eye irritation after contact seek medical advice immediately and show the package insert or the label to the physician.
4.6 Adverse reactions (frequency and seriousness)
Some horses with heavy infections of Onchocerca spp. microfilariae have experienced oedema and pruritis following treatment; such reactions were assumed to be the result of the death of large numbers of microfilariae.
These signs resolve within a few days but symptomatic treatment may be advisable.
In cases of heavy infestations with tapeworms, signs of mild, transient colic and loose stool may be observed. Following administration of Eqvalan Duo, there have been rare reports of inflammation of the mouth, lip and tongue, which results in various clinical signs such as oedema, hypersalivation, erythema, tongue disorder and stomatitis.
These reactions have been transitory in nature, appearing within 1 hour and abating within 24 to 48 hours following administration. In case of severe oral reactions symptomatic treatment is recommended.
4.7 Use during pregnancy, lactation or lay
Studies performed in laboratory animals showed no teratogenic or embryotoxic effect of either ivermectin or praziquantel at the recommended doses during therapy.
Ivermectin-Praziquantel combination can be used after the first three months of gestation and during lactation.
In the absence of clinical data in early pregnancy EQVALAN DUO can only be used in the first three months of gestation according to a risk benefit analysis by the veterinarian.
4.8 Interaction with other medicinal products and other forms of interactions
No data available.
4.9 Amounts to be administered and administration route
The recommended dosage is 200 mcg ivermectin per kilogram of bodyweight and 1mg praziquantel per kilogram of bodyweight corresponding to 1.29 g of paste per 100 kg bodyweight in a single administration.
Bodyweight and dosage should be accurately determined prior to treatment.
For syringes intended to treat horses up to 600 kg and 1100 kg, calibrated markings are provided at 100 kg bodyweight intervals.
For the syringe intended to treat horses up to 750 kg, calibrated markings are provided at 125 kg bodyweight intervals.
The syringe should be adjusted to the calculated dosage by setting the ring on the appropriate place on the plunger.
Directions for use
The product is for oral administration only.
While holding the plunger, turn the knurled ring on the plunger ¼ turn to the left and slide it so the stop ring is at the prescribed weight marking.
Lock the ring in place by turning it ¼ turn to the right in order to bring the two arrows, the one visible on the ring and the one on the plunger rod, into alignment.
Make sure the horse’s mouth contains no feed.
Remove the cover from the tip of the syringe. Insert the syringe tip into the horse’s mouth at the interdental space and deposit the paste on the base of the tongue.
Immediately raise the horse’s head for a few seconds after dosing and ensure that the paste is consumed.
Parasite control Program Veterinary advice should be given on appropriate dosing programs and stock management to achieve adequate parasite control for both tapeworm and roundworm infestations.
4.10 Overdose (symptoms, emergency procedures, antidotes), if necessary
No undesirable effects related to treatment were observed in 2 month old horses treated with EQVALAN DUO, oral paste at up to three times the recommended dose and in adult horses treated at ten times the recommended dose.
Transient decreased food consumption, increased body temperature, salivation and impairment of vision were noticed in horses treated twice with an ivermectin oral paste or once with EQVALAN DUO, oral paste at ten times the recommended dose (i.e., 2 mg/kg b.w.).
All changes disappeared within five days. No antidote has been identified; however, symptomatic therapy may be beneficial.
4.11 Withdrawal period(s)
Meat: 30 days.
Do not use in mares producing milk for human consumption.
5 PHARMACOLOGICAL or IMMUNOLOGICAL PROPERTIES
Pharmacotherapeutic group: Anthelmintics
ATCvet code:
QP54AA51 ivermectin, combinations.
EQVALAN DUO, oral paste is an endectocide containing an association of an anthelmintic active ingredient, ivermectin, and a cestocide active ingredient, praziquantel.
5.1 Pharmacodynamic properties
Ivermectin is a member of the macrocyclic lactone class of endectocides.
Compounds of the class bind selectively and with high affinity to glutamate-gated chloride ion channels which occur in invertebrate nerve and muscle cells.
This leads to an increase in the permeability of the cell membrane to chloride ions with hyperpolarization of the nerve or muscle cell, which results in paralysis and death of the parasite.
Compounds of this class may also interact with other ligand-gated chloride channels, such as those gated by the neurotransmitter gamma-aminobutyric acid (GABA).
The margin of safety for compounds of this class is attributable to the fact that mammals do not have glutamate-gated chloride channels, the macrocyclic lactones have a low affinity for other mammalian ligand-gated chloride channels, and macrocyclic lactones do not readily cross the blood-brain barrier.
Praziquantel is a synthetic isoquinoline-pyrazine derivative with activity against several trematode and cestode parasites.
In vitro and in vivo studies have found that trematodes and cestodes rapidly take up praziquantel within minutes; praziquantel causes tetanic contraction of the parasites' musculature and a rapid vacuolisation of their tegument.
The net effect is that the parasite detaches from the host.
Praziquantel affects membrane permeability in trematodes and cestodes, and influences divalent cation fluxes, particularly calcium ion homeostasis, which is thought to contribute to the rapid muscle contraction and vacuolisation.
The margin of safety for the praziquantel is due to its rapid metabolism and excretion as well as its selective effect on susceptible parasites.
5.2 Pharmacokinetic particulars
After oral administration to horses of the recommended dose of EQVALAN DUO oral paste, praziquantel is rapidly absorbed and excreted, whereas ivermectin is more slowly absorbed and persist during a longer period in the body.
Praziquantel maximum plasma concentrations (of the order of 1 µg/ml) are reached rapidly (approximately in the hour following treatment).
The praziquantel plasma residue depletes rapidly to non-quantifiable levels by 7.5 hours post dose.
Praziquantel is excreted as metabolites in the urine and faeces and the total amount excreted accounts for 31% and 24%, respectively of the administered dose within 24 hours.
Ivermectin maximum plasma concentrations (Cmax: 37.9 ng/ml) are reached in a longer period (tmax: approximately 9 hours after treatment) and levels fell to non detectable / no quantifiable values on or before 28 days after administration.
Faecal excretion is the major pathway of ivermectin elimination in all species studied.
No pharmacological interference between ivermectin and praziquantel was noted.
6 PHARMACEUTICAL PARTICULARS
6.1 List of excipients
Sunset yellow FCF (E110)
Titanium oxide (E171),
Butylhydroxyanisole (E320)
Hydroxypropylcellulose
Hydrogenated castor oil
Glycerol formal
6.2 Major incompatibilities
Not applicable.
6.3 Shelf-life
Shelf life of the veterinary medicinal product as packaged for sale: 3 years Shelf life after first opening the immediate packaging: 2 years
6.4 Special precautions for storage
Store in the original container.
Replace the cap after use.
6.5 Nature and composition of immediate packaging
Immediate package
For syringe intended for the treatment of horsesup to 600 kg, containing 7.74 g of paste:
White polypropylene syringes barrel with a white LDPE cap, a rubber rod tip and a white polypropylene plunger rod, with dose divisions calibrated by body weight with an orange polypropylene stop ring.
For syringes intended for the treatment of horses up to 750 kg and 1100 kg, containing 9.68 g or 14.19 g of paste respectively: White polypropylene syringes barrel with a orange rubber cap, a rubber rod tip and a white polypropylene plunger rod, with dose divisions calibrated by body weight with an orange polypropylene stop ring.
Outer package and sales presentations:
Each syringe is sealed in a transparent polypropylene bag.
Carton box of 1 syringe for oral administration of 7.74g
Carton box of 1 syringe for oral administration of 9.68g
Carton box of 1 syringe for oral administration of 14.19g
Carton box of 50 syringes for oral administration of 7.74g
Carton box of 50 syringes for oral administration of 9.68g
Carton box of 50 syringes for oral administration of 14.19g
Not all pack sizes may be marketed.
6.6 Special precautions for the disposal of unused veterinary medicinal products or waste materials derived from the use of such products
EXTREMELY DANGEROUS FOR FISH AND AQUATIC LIFE.
Do not contaminate surface waters or ditches with product or used syringes.
Any unused product or waste material should be disposed of in accordance with national requirements.
7 MARKETING AUTHORISATION HOLDER
Boehringer Ingelheim Vetmedica GmbH
Binger Strasse 173
55216 Ingelheim am Rhein
Germany
8 MARKETING AUTHORISATION NUMBER(S)
VPA10454/036/001
9 DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 27 May 2005
Date of last renewal: 12 February 2010
10 DATE OF REVISION OF THE TEXT
December 2021
Ivermectin is a semi-synthetic antiparasitic medication derived from avermectins, a class of highly active broad-spectrum antiparasitic agents isolated from the fermentation products of Streptomyces avermitilis.
Ivermectin itself is a mixture of two avermectins, comprising roughly 90% 5-O-demethyl-22,23-dihydroavermectin A1a (22,23-dihydroavermectin B1a) and 10% 5-O-demethyl-25-de(1-methylpropyl)-22,23-dihydro-25-(1-methylethyl) avermectin A1a (22,23-dihydroavermectin B1b).
Pharmacodynamics
Ivermectin is a semisynthetic, anthelminitic agent. It is an avermectin, a group of pentacyclic sixteen-membered lactones (i.e., a macrocyclic lactone disaccharide) derived from the soil bacterium Streptomyces avermitilis. Avermectins are potent and broad-spectrum anti-parasitic agents.
Mechanism of action
Ivermectin binds selectively and with high affinity to glutamate-gated chloride ion channels in invertebrate muscle and nerve cells of the microfilaria.
This binding causes an increase in the permeability of the cell membrane to chloride ions and results in hyperpolarization of the cell, leading to paralysis and death of the parasite.
Ivermectin also is believed to act as an agonist of the neurotransmitter gamma-aminobutyric acid (GABA), thereby disrupting GABA-mediated central nervous system (CNS) neurosynaptic transmission.
Ivermectin may also impair normal intrauterine development of O. volvulus microfilariae and may inhibit their release from the uteri of gravid female worms.
Eqvalan Duo Horse Wormer (Ivermectin, Praziquantel) is an oral paste wormer for horses containing ivermectin and Praziquantel as it's active ingredients. Eqvalan Duo Horse Wormer treats internal parasites, including small and large redworms, lungworms, pinworms and bots in horses.
Active Ingredient: Ivermectin, Praziquantel
Target Species: Horse
Treats and Controls: Large redworms, Small redworms, Pinworms, Lungworms, Ascarids, Stomach worms, Intestinal threadworms, Cutaneous worms, Bots
Withdrawal Time: Withdrawal period for meat and offal is 30 days. Do not use in mares producing milk for human consumption.
Legal Status: POM
Here at Agridirect we have joined forces with DPD to ensure all packages are delivered promptly and safely to you. We ship to all mainland countries within the EU. Deliveries take place Monday to Friday excluding bank holidays. Once your order has been dispatched from our warehouse you will be notified by email. If there is a delay with your order for any reason you will be contacted immediately.
Due to Brexit we are temporarily unable to ship to the UK. Shipping to Northern Ireland will remain in place.
| Ireland (ROI & NI) | EU (Mainland Only) |
| 2-4 Working Days | 4-6 Working Days |
Some products have an extended delivery time, this is noted on the products.
| Country | Orders Under €129 | Orders Over €129 |
| Ireland (ROI & NI) | €7.99 | €0.00 |
| Austria | €39.99 | €32.00 |
| Belgium | €36.99 | €29.00 |
| Czech Republic | €39.99 | €32.00 |
| Denmark | €39.99 | €32.00 |
| Finland | €51.99 | €45.00 |
| France | €36.99 | €29.00 |
| Germany | €36.99 | €29.00 |
| Hungary | €43.99 | €37.00 |
| Italy | €49.99 | €42.00 |
| Luxenburg | €36.99 | €29.00 |
| Netherlands | €36.99 | €29.00 |
| Poland | €36.99 | €29.00 |
| Portugal | €51.99 | €45.00 |
| Slovakia | €43.99 | €37.00 |
| Slovenia | €43.99 | €37.00 |
| Spain | €49.99 | €42.00 |
| Sweden | €49.99 | €42.00 |
A selection of the products we sell are only licensed for sale within the Republic Of Ireland and can not be shipped outside of the country. These products are noted as only being available within the Republic of Ireland on the individual product pages.
There may be an addition charge on certain bulky items. This charge will be clearly marked on an applicable products and will be explained on the checkout page before payment has been made.
We’re sorry your purchase didn’t work out. But don’t worry; we have a great returns policy to help you out.
All purchases can be returned to us within 14 days of delivery and returned goods must be received within 14 days from the date you informed us of the return.
Purchases may be opened for inspection but must not be used and must be repackaged securely in the original packaging if you wish to return it.
If we discover goods have been used or there has been a loss in value of the goods due to damage to the goods, while in your care or whilst being returned to us, we will reduce the amount refunded, which may amount to the full cost of the product, to cover loss of value of goods.
All returns should be complete which includes boxes, manuals and accessories that may have been included with the order.
All returns must be packaged appropriately for shipping, we will not accept responsibility for damages or loss which occur during shipping of a return product.
We accept no responsibility for goods damaged or lost while in transit to us.
We have partnered with DPD to make your returns process easy and secure. simply follow the steps below and bring your package to an official DPD pickup point.
1) All returns must be accompanied with a fully filled out returns form which can be downloaded returns form PDF.
2) To print off your return label click DPD Returns page or visit www.dpd.ie/returns and follow the on-screen instructions. Make sure and use your order number as your reference.
3) Bring your package to a DPD pickup point. To find your nearest drop off point DPD pickup shops.
Once the returned product has been received into our warehouse and been fully inspected a refund will be issued.
If you choose not to use the DPD returns service we recommend that you use a method that can be tracked.
For the return of bulk products please contact us at sales@agridirect.ie
First off, if you have received a damaged electrical product from us, do not plug it in. Any electrical products that are plugged in are deemed ‘as used and accepted’ and are not accepted as returns. All damages must be reported to us via phone or email within 24hours of receipt of goods. Please ensure you check your items upon delivery.
How do I begin the returns process?
If you wish to begin the return process, please email us at sales@agridirect.ie and ensure the following information is included in your email. Your name, phone number, Order id, the item you wish to return, reason for return and if the product is damaged we require photos of the product.
Once you have sent us all required information a member of our team will assess your claim and will contact you as soon as possible. Please hold off on returning products until a member of our team has called you to confirm.
Once the returned product has been returned to us and fully inspected a refund will be issued.
Please Note: A typical timeline for a refund to show in your account is up to 10 working days from the date processed, depending on your bank.
Would you like to send this voucher to the recipient via email?
Yes No