This is a prescription-only medicine (POM).
A valid prescription is required to purchase this product.
Don’t have a prescription? Our team of vets is here to help.
Add to cart as normal and follow the steps. Available in Ireland only.
Volume:
Eprinex Pour-on is designed for use on beef and dairy cattle as well as sheep and goats. It is used for the treatment of and control of gastro-intestinal worms (including inhibited Ostrtagia ostertagia and Cooperia spp.), lungworms, warbles, sucking and biting lice, chorioptic and sarcoptic mange mites in beef and dairy cattle. Eprinex Pour-on boasts a very short withdrawal time for beef cattle and no withdrawal time for dairy cattle. Eprinex provides fast-acting treatment, stopping worm egg output faster than moxidectin and doramectin pour-ons.
Studies
Eprinex has been shown to control sub-clinical parasites and to yield an increase in milk production. In a study of dairy cattle, dairy cattle that had been treated with Eprinex showed a significant and constant increase in milk production. In the first 6 months of lactation, milk production (daily) increased by 0.94kg relative to the placebo group. When Eprinex was give to a herd of cattle diagnosed with Sarcoptic mange, milk production increased by almost 1.5kg per cow per day. (Data courtesy of Boehringer Ingelheim).
In a study of Eprinex treated heifers, calving to conception interval was reduced by an average of 12.9 days. 19.9% more first time heifers were pregnant after the first service.
Active Ingredient: Eprinomectin
Target Species: Cattle, sheep, goats
Administration Method: Pour-On
Treats and Controls: Gastro-intestinal worm, lungworm, sucking and biting lice and mange mites
Withdrawal Time: Cattle: Meat and offal: 15 days. Milk: zero hours. Sheep: Meat and offal: 2 days Milk: zero hours Goats: Meat and offal: 1 day Milk: zero hours
Dosage: 1 ml per 10 kg of bodyweight.
| Body Weight | Dose Volume | Number of full doses per pack: | |||
| 250 ml | 1 Litre | 2.5 Litre | 5 Litre | ||
| 50kg | 5 ml | 50 | 200 | 500 | 1000 |
| 100kg | 10 ml | 25 | 100 | 250 | 500 |
| 150kg | 15 ml | 16 | 66 | 166 | 333 |
| 200kg | 20 ml | 12 | 50 | 125 | 250 |
| 250kg | 25 ml | 10 | 40 | 100 | 200 |
| 300kg | 30 ml | 8 | 33 | 83 | 166 |
| 350kg | 35 ml | 7 | 28 | 71 | 142 |
| 400kg | 40 ml | 6 | 25 | 62 | 125 |
| 450kg | 45 ml | 5 | 22 | 55 | 111 |
| 500kg | 50 ml | 5 | 20 | 50 | 100 |
| 550kg | 55 ml | 4 | 18 | 45 | 90 |
| 600kg | 60 ml | 4 | 16 | 41 | 83 |
Always read the label and all enclosed information for Eprinex Pour-on before administering to animals!
Key Features of Eprinex Pour on
Trials demonstrate that treatment of lactating dairy cows with Eprinex Pour-on can increase milk yield by up to 2 litres per cow per day.
• Treatment with EPRINEX increased milk production:
• Over the whole lactation treated cows produced:
– 63 L more milk
– 2.8 kg more protein
– 2.8 kg more milk fat.
• Treatment cost was recouped within 50 days of calving, with the
treatment investment trebled by the end of lactation.
"The Power To Produce Your Best"
This Product is only licensed for sale in the Republic of Ireland
Click here to Download Data Sheet
Summary of Product Characteristics
1 NAME OF THE VETERINARY MEDICINAL PRODUCT
Eprinex Multi 5 mg/ml pour-on for beef and dairy cattle, sheep and goats
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
Each ml contains:
Active substance:
Eprinomectin........................................................................................... 5.0 mg
Excipients:
Butylhydroxytoluene (E321)..................................................................... 0.1 mg
Alpha-tocopherol……………………………………………………….max. 0.06 mg
For the full list of excipients, see section 6.1.
3 PHARMACEUTICAL FORM
Pour-on solution.
Clear slightly yellow solution.
4 CLINICAL PARTICULARS
4.1 Target Species
Beef and dairy Cattle
Sheep
Goats
4.2 Indications for use, specifying the target species
Treatment of infestation by the following parasites sensitive to eprinomectin:
Cattle
|
PARASITE |
ADULT |
L4 |
Inhibited L4 |
|
Gastrointestinal Roundworms: |
|
|
|
|
Ostertagia spp. |
● |
● |
|
|
O. lyrata |
● |
|
|
|
O. ostertagi |
● |
● |
● |
|
Cooperia spp. |
● |
● |
● |
|
C. oncophora |
● |
● |
|
|
C. punctata |
● |
● |
|
|
C. surnabada C. pectinata |
● ● |
● ● |
|
|
Haemonchus placei |
● |
● |
|
|
Trichostrongylus spp. |
● |
● |
|
|
T. axei |
● |
● |
|
|
T. colubriformis |
● |
● |
|
|
Bunostomum phlebotomum |
● |
● |
|
|
Nematodirus helvetianus |
● |
● |
|
|
Oesophagostomum spp. |
● |
|
|
|
Oesophagostomum radiatum |
● |
● |
|
|
Trichuris spp. |
● |
|
|
|
Lungworm: |
|
|
|
|
Dictyocaulus viviparus |
● |
● |
|
Warbles (parasitic stages)
Hypoderma bovis
Hypoderma lineatum
Mange mites
Chorioptes bovis
Sarcoptes scabiei var. bovis
Lice
Linognathus vituli
Damalinia bovis
Haematopinus eurysternus
Solenopotes capillatus
Flies
Haematobia irritans
PROLONGED ACTIVITY
Applied as recommended, the product prevents reinfestations with:
|
Parasite |
Prolonged Activity |
|
Dictyocaulus viviparus |
Up to 28 days |
|
Ostertagia ostertagi |
Up to 28 days |
|
Oesophagostomum radiatum |
Up to 28 days |
|
Cooperia punctata |
Up to 28 days |
|
Cooperia surnabada Cooperia oncophora |
Up to 28 days Up to 28 days |
|
Nematodirus helvetianus |
Up to 14 days |
|
Trichostrongylus colubriformis |
Up to 21 days |
|
Trichostrongylus axei |
Up to 21 days |
|
Haemonchus placei |
Up to 21 days |
For best results Eprinex Multi should be part of a programme to control both internal and external parasites of cattle based on the epidemiology of these parasites.
Sheep
Gastrointestinal roundworms (adults)
Teladorsagia circumcincta (pinnata/trifurcata)
Haemonchus contortus
Trichostrongylus axei
Trichostrongylus colubriformis
Nematodirus battus
Cooperia curticei
Chabertia ovina
Oesophagostomum venulosum
Lungworm (adult)
Dictyocaulus filaria
Goats
Gastrointestinal roundworms (adult)
Teladorsagia circumcincta (pinnata/trifurcata)
Haemonchus contortus
Trichostrongylus axei
Trichostrongylus colubriformis
Nematodirus battus
Cooperia curticei
Oesophagotomum venulosum
Lungworm (adult)
Dictyocaulus filaria
4.3 Contraindications
This product is formulated only for topical application to cattle, sheep and goatsincluding lactating dairy animals.
Do not use in other animal species.
Do not use in cases of hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings for each target species
For effective use, the product should not be applied to areas of the backline covered with mud or manure.
The product should be applied only on healthy skin.
In cattle,
rainfall before, during or after the application of the product, has been shown to have no impact on its efficacy.
It also has been demonstrated that haircoat length has no impact on the product’s efficacy.
The effect of rainfall and haircoat length on efficacy has not been evaluated in sheep and goats.
For cattle, in order to limit cross-transfer of eprinomectin, treated animals may be separated from untreated animals. Non-compliance withthis recommendation may lead to residue violations in untreated animals.
Care should be taken to avoid the following practices because they increase the risk of development of resistance and could ultimately result in ineffective therapy:
-Too frequent and repeated use of anthelmintics from the same class, over an extended period of time.
-Underdosing, which may be due to underestimation of bodyweight, misadministration of the product, or lack of calibration ofthe dosing device (if any).
Suspected clinical cases of resistance to anthelmintics should be further investigated using appropriate tests (e.g. Faecal Egg Count Reduction Test). Where the results of the test(s) strongly suggest resistance to a particular anthelmintic, an anthelmintic belonging to another pharmacological class and having a different mode of action should be used.
To date no resistance to eprinomectin (a macrocyclic lactone) has been reported in cattle and sheep while resistance to eprinomectin has been reported in goats within the EU.
However resistance to other macrocyclic lactones has been reported in nematode populations in cattle, sheep and goats within the EU, which may be associated with side-resistance to eprinomectin.
Therefore, use of this product should be based on local (regional, farm) epidemiological information about susceptibility of nematodes and recommendations on how to limit further selection for resistance to anthelmintics.
While mite and louse numbers decline rapidly following treatment, due to the feeding habits of some mites, in some cases several weeks may be required for complete eradication.
4.5 Special precautions for use
Special precautions for use in animals
For external use only.
Not to be used in other species; avermectins can cause fatalities in dogs, especially Collies, Old English Sheepdogs and related breeds and crosses, and also in turtles/tortoises.
To avoid secondary reactions due to the death of Hypodermalarvae in the oesophagus or in the spine, it is recommended to administer the product at the end of warble fly activity and before the larvae reach their resting sites.
Special precautions to be taken by the person administering the veterinary medicinal product to animal.
Do not smoke, eat or drink while handling the product.
Wash hands after use.
This product may be irritating to skin and eyes. Avoid contact with eyes and skin
People with known hypersensitivity to the active substance or to any of the excipients should avoid contact with the product.
Operators should wear rubber gloves, boots and waterproof coat when applying the product.
If clothing become contaminated, remove as soon as possible and launder before re-use.
If accidental skin contact occurs, wash the affected area immediately with soap and water.
Should accidental eye exposure occur, flush eyes immediately with plenty of clean water.
Should irritation persist, seek medical advice.
Do not ingest.
In case of accidental ingestion, rinse out mouth thoroughly with water, seek medical advice immediately and show the package insert or the label to the physician.
Other precautions
Eprinomectin is very toxic to dung fauna and aquatic organisms, is persistent in soils and may accumulate in sediments. The risk to aquatic ecosystems and dung fauna can be reduced by avoiding repeated use of eprinomectin (and products of the same anthelmintic class).
In order to reduce the risk to aquatic ecosystems, treated animals should not have direct access to water bodies for a minimum of two weeks after treatment.
4.6 Adverse reactions (frequency and seriousness)
- In very rare cases, pruritus and alopecia have been observed after the use of the veterinary medicinal product
- The frequency of adverse reactions is defined using the following convention:
4.7 Use during pregnancy, lactation or lay
Laboratory studies (rat, rabbit) have not produced any evidence of a teratogenic or embryotoxic effects due to the use of eprinomectin at therapeutic doses.
Laboratory studies in cattle have not produced any evidence of a teratogenic or foetotoxic effect at the recommended therapeutic dose.
The product can be used in dairy cattle during pregnancy and lactation.
The safety of eprinomectin during pregnancy in sheep and goats has not been tested. Use only according to the benefit/risk assessment of the responsible veterinarian in these species.
4.8 Interaction with other medicinal products and other forms of interactions
No interactions with other medicines and no other forms of interactions are known.
Since eprinomectin binds extensively to plasmatic proteins, this should be taken into account if it is used in association with other molecules having the same characteristics.
4.9 Amounts to be administered and administration route
Pour-on use
To ensure administration of a correct dose, bodyweight should be determined as accurately as possible; accuracy of the dosing device should be checked. If animals are to be treated collectively rather than individually, they should be grouped according to their bodyweight and dosed accordingly, in order to avoid under- and over- dosing.
The product should be applied topically by pouring along the backline in a narrow strip extending from the withers to the tailhead.
Cattle:
Administer by topical application at the dose rate of 0.5 mg eprinomectin per kg bodyweight, corresponding to the recommended dose rate of 1 ml per 10 kg bodyweight.
Sheep and goats:
Administer by topical application at the dose rate of 1.0 mg eprinomectin per kg bodyweight, corresponding to the recommended dose rate of 2 ml per 10 kg bodyweight.
When administering the product along the backline, part the fleece/coat and place applicator nozzle or bottle spout against the skin.
Method of administration
For 250 ml and 1 litre bottles :
For 2.5 and 5 litre
No signs of toxicity were observed when 8 - week old calves were treated at up to 5 times the therapeutic dose (2.5 mg eprinomectin/kg bodyweight.) 3 times at 7 - day intervals.
One calf treated once at 10 times the therapeutic dose (5 mg/kg bodyweight.) in the tolerance study showed transient mydriasis. There were no other adverse reactions to the treatment.
No signs of toxicity were observed when 17 - week old sheep were treated at doses up to 5 times the therapeutic dose (5 mg eprinomectin/kg bodyweight) 3 times at 14 - day intervals.
No antidote has been identified.
4.11 Withdrawal period(s)
Cattle:
Meat and offal: 15 days.
Milk: zero hours.
Sheep:
Meat and offal: 2 days
Milk: zero hours
Goats:
Meat and offal: 1 day
Milk: zero hours
5 PHARMACOLOGICAL or IMMUNOLOGICAL PROPERTIES
Pharmacotherapeutic group:
Antiparasitic products, Avermectins.
ATCvet code: QP54AA04
5.1 Pharmacodynamic properties
Eprinomectin is a member of the macrocyclic lactone class of endectocides.
Compounds of the class bind selectively and with high affinity to glutamate-gated chloride ion channels which occur in invertebrate nerve or muscle cells.
This leads to an increase in the permeability of the cell membrane to chloride ions with hyperpolarization of the nerve or muscle cell, resulting in paralysis and death of the parasite.
Compounds of this class may also interact with other ligand-gated chloride channels, such as those gated by the neurotransmitter gamma-aminobutyric acid (GABA).
The margin of safety for compounds of this class is attributable to the fact that mammals do not have glutamate-gated chloride channels; the macrocyclic lactones have a low affinity for other mammalian ligand-gated chloride channels, and they do not readily cross the blood-brain barrier.
5.2 Pharmacokinetic particulars
Eprinomectin is bound extensively to plasma proteins (99 %).
Pharmacokinetic studies have been conducted in lactating and non-lactating animals, administered topically at a single dosage of 0.5 mg/kg body weight in cattle and at 1 mg/kg bodyweight in sheep and goats.
For cattle, results from two representative studies found mean peak plasma concentrations of 9.7 and 43.8 ng/ml that were observed at 4.8 and 2.0 days post dose. The corresponding elimination half-lives in plasma were 5.2 and 2.0 days, and mean area-under-the-curve values of 124 and 241 ng*day/ml.
Eprinomectin is not extensively metabolized in cattle following topical administration. Faeces was the major route of elimination of the drug in beef cattle and dairy cows.
For sheep, a mean peak plasma concentration (Cmax) of 6.20 ng/ml was observed following a topical dose of 1mg/kg. The half-life in plasma was 6.4 days with mean area under the curve (AUClast) value of 48.8 ng*day/ml.
For goats, peak mean plasma concentrations ranging from 3 to 13.1 ng/ml were observed from day 1 to day 2 post dose. The half life in plasma ranged from less than one day to 3 days with area under the curve mean values ranging from 15.7 to 39.1 ng-day/ml.
An in vitro microsomal metabolism study was conducted using liver microsomes isolated from cattle, sheep and goats. It showed that the differences in pharmacokinetics observed between cattle, sheep and goats do not result from differences in the rate or extent of metabolism but suggests more complete absorption of eprinomectin by cattle.
Environmental properties
See section 4.5 (other precautions).Like other macrocyclic lactones, eprinomectin has the potential to adversely affect non-target organisms. Following treatment, excretion of potentially toxic levels of eprinomectin may take place over a period of several weeks. Faeces containing eprinomectin excreted onto pasture by treated animals may reduce the abundance of dung feeding organisms which may impact on the dung degradation.
6 PHARMACEUTICAL PARTICULARS
6.1 List of excipients
Butylhydroxytoluene (E321)
Propylene glycol dicaprylocaprate Alpha-tocopherol
6.2 Major incompatibilities
In the absence of compatibility studies, this veterinary medicinal product must not be mixed with other veterinary medicinal products.
6.3 Shelf-life
Shelf life of the veterinary medicinal product as packaged for sale: 3 years. Shelf life after first opening the immediate packaging: see expiry date.
6.4 Special precautions for storage
Keep the container in the outer carton in order to protect from light.
This veterinary medicinal product does not require any special temperature storage conditions.Store container upright.
6.5 Nature and composition of immediate packaging
250 ml and 1 L HDPE bottle
2.5 and 5 L HDPE back pack
Sealed foil and tamper evident
HDPE screw cap with polypropylene liner
250 ml bottle with 2 measuring devices of 25 ml (1 for cattle, 1 for sheep/goat)
1L bottle with 2 measuring devices (1 of 60 ml for cattle, 1 of 25 ml for sheep/goat)
2.5L back-pack with a dispensing cap
5L back-pack with a dispensing cap
One bottle or one back-pack per cardboard box.
The 2.5 litre and 5 litre back-packs are designed for use with a suitable automatic dispensing gun. Not all pack sizes may be marketed.
6.6 Special precautions for the disposal of unused veterinary medicinal products or waste materials derived from the use of such products
Any unused veterinary medicinal product or waste materials derived from such veterinary medicinal product should be disposed of in accordance with local requirements.
Do not contaminate surface waters or ditches with the product or used container.
7 MARKETING AUTHORISATION HOLDER
Boehringer Ingelheim Vetmedica GmbH
Binger Strasse 173
55216 Ingelheim am Rhein
Germany
10 DATE OF REVISION OF THE TEXT
August 2019
Sheep Pour-ons and spot-ons
Pour-ons and spot-ons need to be applied accurately and each manufacturer may recommend subtle differences. Use appropriate and calibrated guns, always clean with warm soapy water and then rinse after use. Store in a safe dry place. When treating sheep with these products, make sure they are applied along the back line. If placed to one side, the product will not spread evenly around the body. No pour-on or spot-on is effective against sheep scab.
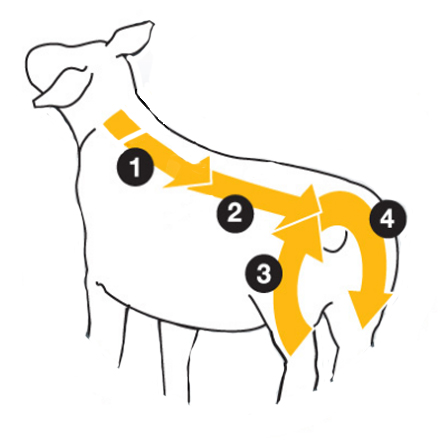
The Four Stroke Method: The four stroke method ensures the pour-on is applied evenly by applying a quarter of the application at a time in four simple strokes.
1) 1/4 applied from between the shoulders to middle of back
2) 1/4 applied from middle of back to top of animals tail
3) 1/4 applied from one side of the animals tail down the crutch
4) 1/4 applied from other side of tail down the other crutch
Cattle Pour-ons
These should be applied along the length of the flattest part of the animal’s back, from the withers to the tail head.
In general, animals should not be treated when the hair is wet or if rain is anticipated within two hours of treatment. However, some products are waterproof and can be used on wet animals.
Areas of damaged skin should be avoided, as should areas contaminated with mud or manure

What is Eprinomectin?
Eprinomectin is a semi-synthetic compound of the avermectin family, intended for the treatment of internal and external parasites in cattle and in lactating cows.
Eprinomectin is a mixture of two homologues, eprinomectin B1a (90%) and eprinomectin B1b (10%), which differ by a methylene group in the C25.
The recommended dosage regimen is a single dose of 0.5 mg/kg bw (0.1 ml/10 kg bw) applied topically along the midline of the animal's back.
How does Eprinomectin work?
The precise mode of action of eprinomectin remains unknown, despite extensive investigations with a variety of compounds from the same class.
Eprinex Pour-on is designed for use on beef and dairy cattle as well as sheep and goats. It is used for the treatment of and control of gastro-intestinal worms (including inhibited Ostrtagia ostertagia and Cooperia spp.), lungworms, warbles, sucking and biting lice, chorioptic and sarcoptic mange mites in beef and dairy cattle.
Active Ingredient: Eprinomectin
Target Species: Cattle, sheep, goats
Treats and Controls: Gastro-intestinal worm, lungworm, sucking and biting lice and mange mites
Withdrawal Time: Cattle: Meat and offal: 15 days. Milk: zero hours. Sheep: Meat and offal: 2 days Milk: zero hours Goats: Meat and offal: 1 day Milk: zero hours
Legal Status: POM
Here at Agridirect we have joined forces with DPD to ensure all packages are delivered promptly and safely to you. We ship to all mainland countries within the EU. Deliveries take place Monday to Friday excluding bank holidays. Once your order has been dispatched from our warehouse you will be notified by email. If there is a delay with your order for any reason you will be contacted immediately.
Due to Brexit we are temporarily unable to ship to the UK. Shipping to Northern Ireland will remain in place.
| Ireland (ROI & NI) | EU (Mainland Only) |
| 2-4 Working Days | 4-6 Working Days |
Some products have an extended delivery time, this is noted on the products.
| Country | Orders Under €129 | Orders Over €129 |
| Ireland (ROI & NI) | €7.99 | €0.00 |
| Austria | €39.99 | €32.00 |
| Belgium | €36.99 | €29.00 |
| Czech Republic | €39.99 | €32.00 |
| Denmark | €39.99 | €32.00 |
| Finland | €51.99 | €45.00 |
| France | €36.99 | €29.00 |
| Germany | €36.99 | €29.00 |
| Hungary | €43.99 | €37.00 |
| Italy | €49.99 | €42.00 |
| Luxenburg | €36.99 | €29.00 |
| Netherlands | €36.99 | €29.00 |
| Poland | €36.99 | €29.00 |
| Portugal | €51.99 | €45.00 |
| Slovakia | €43.99 | €37.00 |
| Slovenia | €43.99 | €37.00 |
| Spain | €49.99 | €42.00 |
| Sweden | €49.99 | €42.00 |
A selection of the products we sell are only licensed for sale within the Republic Of Ireland and can not be shipped outside of the country. These products are noted as only being available within the Republic of Ireland on the individual product pages.
There may be an addition charge on certain bulky items. This charge will be clearly marked on an applicable products and will be explained on the checkout page before payment has been made.
We’re sorry your purchase didn’t work out. But don’t worry; we have a great returns policy to help you out.
All purchases can be returned to us within 14 days of delivery and returned goods must be received within 14 days from the date you informed us of the return.
Purchases may be opened for inspection but must not be used and must be repackaged securely in the original packaging if you wish to return it.
If we discover goods have been used or there has been a loss in value of the goods due to damage to the goods, while in your care or whilst being returned to us, we will reduce the amount refunded, which may amount to the full cost of the product, to cover loss of value of goods.
All returns should be complete which includes boxes, manuals and accessories that may have been included with the order.
All returns must be packaged appropriately for shipping, we will not accept responsibility for damages or loss which occur during shipping of a return product.
We accept no responsibility for goods damaged or lost while in transit to us.
We have partnered with DPD to make your returns process easy and secure. simply follow the steps below and bring your package to an official DPD pickup point.
1) All returns must be accompanied with a fully filled out returns form which can be downloaded returns form PDF.
2) To print off your return label click DPD Returns page or visit www.dpd.ie/returns and follow the on-screen instructions. Make sure and use your order number as your reference.
3) Bring your package to a DPD pickup point. To find your nearest drop off point DPD pickup shops.
Once the returned product has been received into our warehouse and been fully inspected a refund will be issued.
If you choose not to use the DPD returns service we recommend that you use a method that can be tracked.
For the return of bulk products please contact us at sales@agridirect.ie
First off, if you have received a damaged electrical product from us, do not plug it in. Any electrical products that are plugged in are deemed ‘as used and accepted’ and are not accepted as returns. All damages must be reported to us via phone or email within 24hours of receipt of goods. Please ensure you check your items upon delivery.
How do I begin the returns process?
If you wish to begin the return process, please email us at sales@agridirect.ie and ensure the following information is included in your email. Your name, phone number, Order id, the item you wish to return, reason for return and if the product is damaged we require photos of the product.
Once you have sent us all required information a member of our team will assess your claim and will contact you as soon as possible. Please hold off on returning products until a member of our team has called you to confirm.
Once the returned product has been returned to us and fully inspected a refund will be issued.
Please Note: A typical timeline for a refund to show in your account is up to 10 working days from the date processed, depending on your bank.
Would you like to send this voucher to the recipient via email?
Yes No