This is a prescription-only medicine (POM).
A valid prescription is required to purchase this product.
Don’t have a prescription? Our team of vets is here to help.
Add to cart as normal and follow the steps. Available in Ireland only.
Volume:
Active Ingredient: Fenbendazole
Target Species: Sheep
Treats and Controls: Gastrointestinal roundworms, lungworms
Administration Method: Oral Drench
Withdrawal Time: 28 days for animals intended for meat and offal, not permitted for use on animals producing milk for human consumption.
Dosage: 2 ml per 10 kg of bodyweight
| Bodyweight in kg | Dose in ml | Doses per 1 ltr bottle | Doses per 2.5 ltr bottle |
| 10 | 2 | 500 | 1250 |
| 20 | 4 | 250 | 625 |
| 30 | 6 | 166 | 415 |
| 40 | 8 | 125 | 312 |
| 50 | 10 | 100 | 250 |
| 60 | 12 | 83 | 207 |
| 70 | 14 | 71 | 177 |
Always read the label and all enclosed information for Fenbenor 2.5% before administering to animals!
Signs and effects of infected livestock |
|
Infection: Gut Worm Symptoms: Diarrhoea, decreased appetite, loss of weight Effects: Gutworm can cause severe damage to the stomach and small intestine which will cause parasitic gastroenteritis, this will not only negatively affect the health of the animal but will affect the profitability for the farmer. |
|
Infection: Lungworm Symptoms: Short, sharp cough that becomes worse with exercise, in severe cases the animal will have obvious difficulty breathing. Effects: Lungworm infections cause a high susceptibility to respiratory viruses and bacteria. Infected cattle are prone to contracting severe bronchial pneumonia which if left untreated can lead to death |
Key Features of Fenbenor 2.5%
This product is only licensed for sale within the Republic of Ireland
Click here to Download Data Sheet
Irish Medicines Board
Summary of Product Characteristics
1 NAME OF THE VETERINARY MEDICINAL PRODUCT
Fenbenor 2.5% w/v Oral Drench.
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
For a full list of excipients see section 6.1.
Each ml contains:
Active substance
Fenbendazole 25.0 mg
Excipients
Methyl Parahydroxybenzoate (E218) 2.0 mg
Propyl Parahydroxybenzoate (E216) 0.2 mg
SodiumMetabisulphite 1.0 mg
3 PHARMACEUTICAL FORM
Oral suspension.
A white to off-white suspension.
4 CLINICAL PARTICULARS
4.1 Target Species
Sheep.
4.2 Indications for use, specifying the target species
Fenbenor 2.5% w/v Drench is a broad spectrum anthelmintic for control of benzimidazole susceptible mature and
developing immature forms of the following nematodes of the gastrointestinal and respiratory tracts of sheep.
For the treatment of sheep infected with:
Haemonchus spp.
Ostertagia spp.
Trichostrongylus spp.
Cooperia spp.
Nematodirus spp.
Bunostomum spp.
Trichuris spp.
Strongyloides spp.
Oesophagostomum spp.
Dictyocaulus spp
Moniezia spp.
4.3 Contraindications
Do not use in animals with known hypersensitivity to the active ingredient.
4.4 Special warnings for each target species
When dosing sheep, care must be taken not to damage the mouth or pharyngeal region with drenching equipment.
As with other anthelmintics, veterinary advice should be sought on appropriate dosing programmes and stock management to achieve adequate parasite control and reduce the likelihood of anthelmintic resistance developing.
If the product does not achieve the desired clinical effect, other diseases, nutritional disturbances or anthelmintic resistance may be involved.
4.5 Special precautions for use
Special precautions for use in animals
None.
Special precautions to be taken by the person administering the veterinary medicinal product to animals
Estimate bodyweight carefully.
Use only properly calibrated dosing equipment.
4.6 Adverse reactions (frequency and seriousness)
None known.
4.7 Use during pregnancy, lactation or lay
Sheep have been reported to be sensitive to benzimidazoles during the first quarter of gestation.
4.8 Interaction with other medicinal products and other forms of interaction
None known.
4.9 Amounts to be administered and administration route
For oral administration in sheep.
Shake well before use.
The recommended treatment dose is 5 mg Fenbendazole per kg bodyweight.
Practical dosage recommendations are as follows:
Bodyweight (kg) Dose (ml) Number of doses per pack
10 2.0 500
50 10.0 100
4.10 Overdose (symptoms, emergency procedures, antidotes), if necessary
Not applicable.
4.11 Withdrawal Period(s)
Sheep must not be slaughtered for human consumption during treatment or for 28 days thereafter.
This product should not be used in ewes producing milk for human consumption.
5 PHARMACOLOGICAL or IMMUNOLOGICAL PROPERTIES
Pharmacotherapeutic Group:
Anthelmintic, fenbendazole,
ATCvet Code:
QP52AC13.
Summary presentation of the active ingredient
Fenbendazole is an anthelmintic belonging to the benzimidazole group which acts by blocking fumarate reductase which results in the inhibition of the formation of adenosine triphosphate (involved in mitochondrial energy).
5.1 Pharmacodynamic properties
Fenbendazole, like many benzimidazoles, blocks fumarate reductase which results in the inhibition of the formation of
adenosine triphosphate (involved in mitochondrial energy).
There is also evidence that it inhibits glucose uptake and therefore increases glycogen utilization and depletes the worm’s glycogen reserves.
The overall effect of this action is to effectively starve the parasite to death. Furthermore this action results in the detachment of the parasites but in the case of intestinal helminths this detachment does not result in loss of contact with the drug whereas in the case of the liver fluke such detachment would reduce such contact.
This probably explains its limited effect on the liver fluke and the good effect on intestinal helminths.
5.2 Pharmacokinetic properties
Fenbendazole is poorly soluble in water and consequently is poorly absorbed; something which is reflected in the relatively low plasma levels.
The scheme for the known metabolic pathways is given by Short, Flory, Hsieh and Barker (1988) together with the relative rates of breakdown in various species.
The main breakdown products are the sulphoxide (oxfendazole) and sulphone.
6 PHARMACEUTICAL PARTICULARS
6.1 List of excipients
Polysorbate 80
Sodium metabisulphite
Methyl Parahydroxybenzoate (E218)
Propyl Parahydroxybenzoate (E216)
Sodium Citrate
Citric Acid
Simeticone emulsion
Xanthan Gum
PurifiedWater
6.2 Incompatibilities
None known.
6.3 Shelf-life
Shelf life of the veterinary medicinal product as packaged for sale: 3 years.
6.4 Special precautions for storage
Do not store above 25oC.
Do not freeze.
6.5 Nature and composition of immediate packaging
1 L (jerrican, flat bottom flexi), 2.5 L (jerrican, back pack), 5 L (jerrican)
HDPE white rigid containers closed with a HDPP screw cap with a wood pulp PVDC liner.
Not all pack sizes may be marketed.
6.6 Special precautions for the disposal of unused veterinary medicinal products or waste materials
Empty containers must be rinsed with water before disposal.
Dispose of used containers safely.
Do not contaminate ponds, waterways or ditches with product or used containers.
Unused product or waste material should be disposed of in accordance with current practice for pharmaceutical waste under national waste disposal regulations
7 MARKETING AUTHORISATION HOLDER
Pharvet (Ireland) Ltd.,
29 Cookstown Industrial Estate,
Dublin 24,
Ireland.
8 MARKETING AUTHORISATION NUMBER(S)
VPA: 10462/002/002
9 DATE OF THE FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
3rd August 2007
10 DATE OF REVISION OF THE TEXT
October 2012
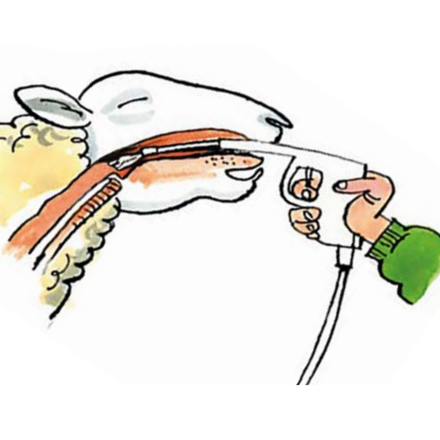
Drench correctly
Sheep Oral Drench
The drenching technique is a vital part of ensuring that the wormer does its job effectively.
Make sure the sheep are properly restrained and cannot leap around when they are being drenched so they swallow the whole amount.
Sheep can also suffer serious injury, or even death, if they are unrestrained and the gun penetrates the tissues at the back of the mouth.
Place a hand under the head and tilt slightly to the side.
Slot the nozzle in the gap between molar and incisor teeth and then over the back of the tongue.
If the wormer is just put into the mouth, it will bypass the rumen as it escapes down the oesophageal groove and will be less effective.
This is particularly important for white (BZ) drenches.
Dosing Weigh – do not guess Underestimating the weight of sheep is a common cause of underdosing. Select and weigh the biggest sheep in the group to determine the correct dose. If there is a wide range of weights, consider splitting the group, then weigh the heaviest in each section. Do not forget to check that the weigh crate is accurate before starting!
Calibrate and maintain the drench gun
Always check the gun is delivering the right amount before you drench. Remove the plunger from a 10 ml syringe, put a thumb over the end and squirt the dose into it, making sure there are no air bubbles left. Adjust the gun until the dose delivered is correct. Drenching guns should also be well maintained and replaced regularly. Clean with warm soapy water after use and check springs and tubes to make sure there are no kinks that will form air bubbles.
Withholding food
Research has shown that the efficacy of the white (BZ) and clear (AV) drenches can be improved by withholding food for 12–24 hours before treatment. It is not advised to deprive heavily pregnant ewes of food, so if you treat this class of stock with anthelmintics, you may wish to use yellow drenches (LV) because their efficacy is less dependent on rumen fill.
Storage
Wormers should be stored securely, away from direct sunlight at 4–25°C. Check the use-by date and, once open, use within the time shown on the packaging. Shake white (BZ) products well before use.
Active Ingredient: Fenbendazole
Target Species: Sheep
Treats and Controls: Gastrointestinal roundworms, lungworms
Withdrawal Time: 28 days for animals intended for meat and offal, not permitted for use on animals producing milk for human consumption.
Legal Status: POM
Here at Agridirect we have joined forces with DPD to ensure all packages are delivered promptly and safely to you. We ship to all mainland countries within the EU. Deliveries take place Monday to Friday excluding bank holidays. Once your order has been dispatched from our warehouse you will be notified by email. If there is a delay with your order for any reason you will be contacted immediately.
Due to Brexit we are temporarily unable to ship to the UK. Shipping to Northern Ireland will remain in place.
| Ireland (ROI & NI) | EU (Mainland Only) |
| 2-4 Working Days | 4-6 Working Days |
Some products have an extended delivery time, this is noted on the products.
| Country | Orders Under €129 | Orders Over €129 |
| Ireland (ROI & NI) | €7.99 | €0.00 |
| Austria | €39.99 | €32.00 |
| Belgium | €36.99 | €29.00 |
| Czech Republic | €39.99 | €32.00 |
| Denmark | €39.99 | €32.00 |
| Finland | €51.99 | €45.00 |
| France | €36.99 | €29.00 |
| Germany | €36.99 | €29.00 |
| Hungary | €43.99 | €37.00 |
| Italy | €49.99 | €42.00 |
| Luxenburg | €36.99 | €29.00 |
| Netherlands | €36.99 | €29.00 |
| Poland | €36.99 | €29.00 |
| Portugal | €51.99 | €45.00 |
| Slovakia | €43.99 | €37.00 |
| Slovenia | €43.99 | €37.00 |
| Spain | €49.99 | €42.00 |
| Sweden | €49.99 | €42.00 |
A selection of the products we sell are only licensed for sale within the Republic Of Ireland and can not be shipped outside of the country. These products are noted as only being available within the Republic of Ireland on the individual product pages.
There may be an addition charge on certain bulky items. This charge will be clearly marked on an applicable products and will be explained on the checkout page before payment has been made.
We’re sorry your purchase didn’t work out. But don’t worry; we have a great returns policy to help you out.
All purchases can be returned to us within 14 days of delivery and returned goods must be received within 14 days from the date you informed us of the return.
Purchases may be opened for inspection but must not be used and must be repackaged securely in the original packaging if you wish to return it.
If we discover goods have been used or there has been a loss in value of the goods due to damage to the goods, while in your care or whilst being returned to us, we will reduce the amount refunded, which may amount to the full cost of the product, to cover loss of value of goods.
All returns should be complete which includes boxes, manuals and accessories that may have been included with the order.
All returns must be packaged appropriately for shipping, we will not accept responsibility for damages or loss which occur during shipping of a return product.
We accept no responsibility for goods damaged or lost while in transit to us.
We have partnered with DPD to make your returns process easy and secure. simply follow the steps below and bring your package to an official DPD pickup point.
1) All returns must be accompanied with a fully filled out returns form which can be downloaded returns form PDF.
2) To print off your return label click DPD Returns page or visit www.dpd.ie/returns and follow the on-screen instructions. Make sure and use your order number as your reference.
3) Bring your package to a DPD pickup point. To find your nearest drop off point DPD pickup shops.
Once the returned product has been received into our warehouse and been fully inspected a refund will be issued.
If you choose not to use the DPD returns service we recommend that you use a method that can be tracked.
For the return of bulk products please contact us at sales@agridirect.ie
First off, if you have received a damaged electrical product from us, do not plug it in. Any electrical products that are plugged in are deemed ‘as used and accepted’ and are not accepted as returns. All damages must be reported to us via phone or email within 24hours of receipt of goods. Please ensure you check your items upon delivery.
How do I begin the returns process?
If you wish to begin the return process, please email us at sales@agridirect.ie and ensure the following information is included in your email. Your name, phone number, Order id, the item you wish to return, reason for return and if the product is damaged we require photos of the product.
Once you have sent us all required information a member of our team will assess your claim and will contact you as soon as possible. Please hold off on returning products until a member of our team has called you to confirm.
Once the returned product has been returned to us and fully inspected a refund will be issued.
Please Note: A typical timeline for a refund to show in your account is up to 10 working days from the date processed, depending on your bank.
Would you like to send this voucher to the recipient via email?
Yes No